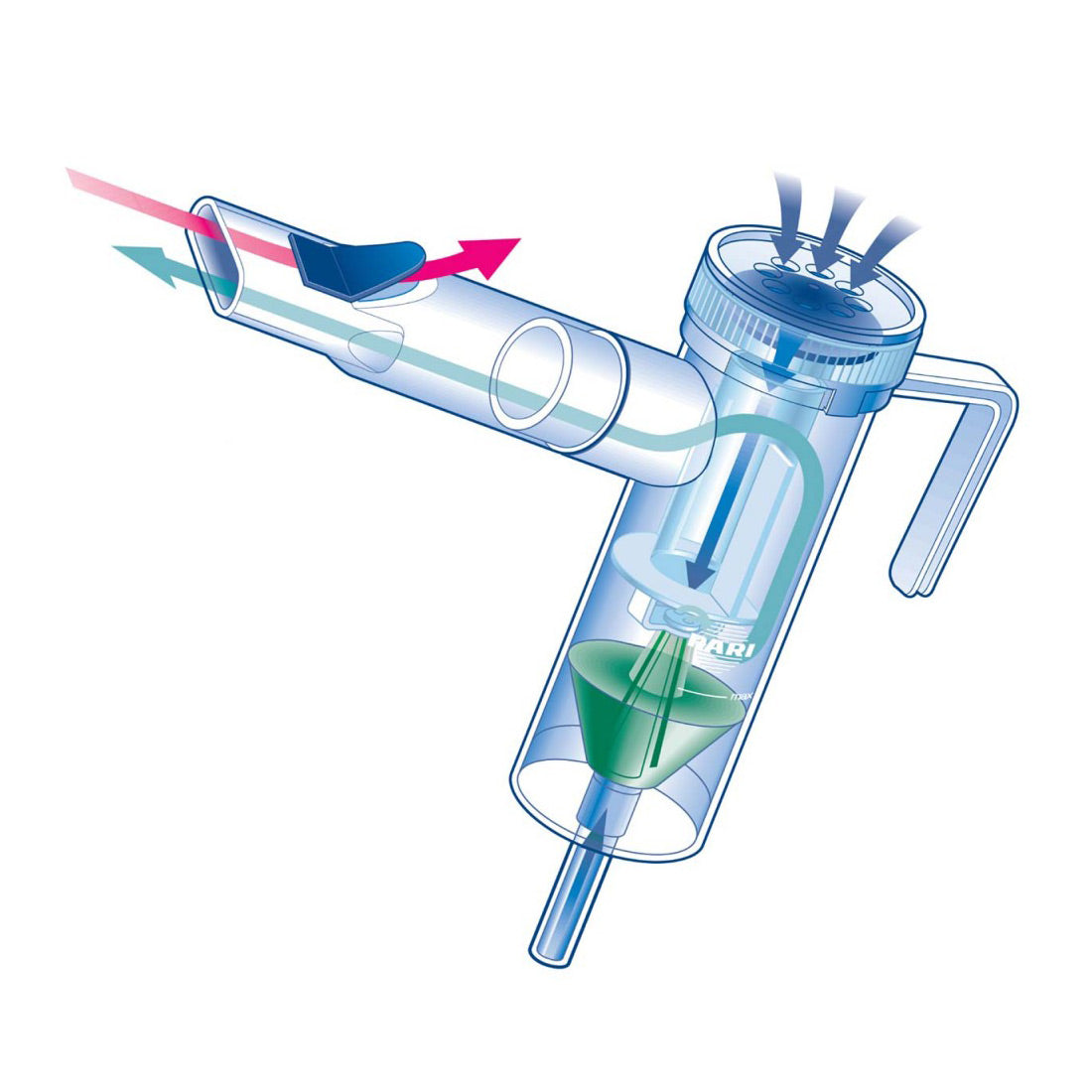
Sunovion Pharmaceuticals Inc. - Sunovion Announces Lonhala™ Magnair™ (glycopyrrolate) Inhalation Solution Now Available in the U.S. for the Treatment of COPD

Sunovion Pharmaceuticals Inc. - Sunovion Receives FDA Approval for Lonhala™ Magnair™ Inhalation Solution to Treat COPD

Mario Cortina - Senior Multi-Specialty Portfolio Specialist - Sumitomo Pharma America (formerly Sunovion Pharmaceuticals | LinkedIn

Sunovion Receives FDA Approval for Lonhala™ Magnair™ Inhalation Solution to Treat COPD | Business Wire